AUD 33.99 – AUD 36.99Price range: AUD 33.99 through AUD 36.99
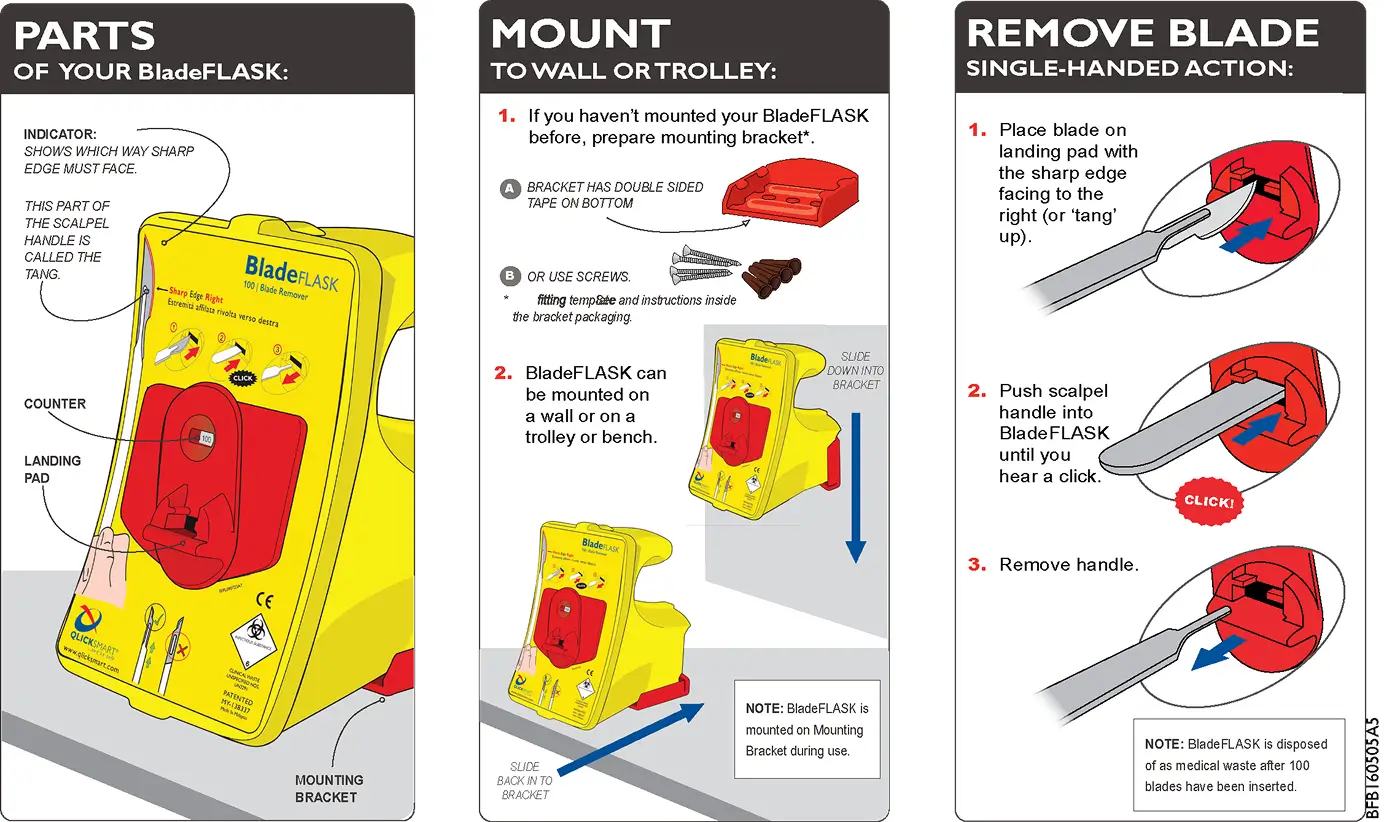
BladeFLASK is simple to use with a quick “push, click, and pull” action to remove and contain used scalpel blades.
Please note that the BladeFLASK is currently available to be purchased online in Australia only. If you’re outside of Australia, please contact us to find a distributor in your area.



The BladeFLASK is a single-handed blade remover engineered to improve sharps safety in any clinical environment. With its automatic containment system, it provides a fast, safe, and sterile way to dispose of used scalpel blades without the need for two hands or additional tools.
Features
Features
Designed for One-Handed Scalpel Blade Removal
Specs
Technical Specifications

Qlicksmart
Mounting Bracket
Guide
How to Use
BladeFLASK
Resources
Resources

Product Brochure

Note: The colour of the product is dependent on your region’s regulations.
RED – USA, Central, and South America markets only
YELLOW – Oceania, Asia, Europe, Africa, and Canada markets only
FAQs
Frequently Asked Questions About BladeFLASK

When should I replace my BladeFlask?
The BladeFLASK shut-off automatically after approximately 100 blades are removed, and then must be disposed and replaced. Please check the count-down indicator which counts down from 100 to 0, then ‘FULL’ to see when you will need to replace the BladeFLASK.
How do I dispose of the BladeFLASK?
Once the BladeFLASK has shut-off, it must be disposed of as medical waste. The BladeFLASK is a puncture-proof sharps container, so you do not need to put it into another sharps container. Please follow your organisation’s standard procedures for disposal of medical waste.
Please follow your organisation’s infection control guidelines on the disposal time-frame for used BladeFLASK, regardless of whether the unit has shut off. The BladeFLASK securely contains blades within the unit regardless of whether the unit has shut off, and has been tested in accordance with the provisions of the United Nations Transport of Dangerous Goods, Chapter 6.
Why do I need a Mounting Bracket?
The Mounting Bracket helps to position the BladeFLASK so that it can remove blades single-handedly. The Mounting Bracket also allows the BladeFLASK to be mounted on a vertical or horizontal surface (eg on a wall or bench), so that blades can be removed at point of use.
How do I install the Mounting Bracket?
You can affix the Mounting Bracket via the industrial-strength double-sided tape, or by screws and lugs which are included in the packaging. A fitting template has been included in the Mounting Bracket packaging to indicate the clearance required for easy installation.
When should I install a new Mounting Bracket?
You can re-use the Mounting Bracket until the current bracket starts to become loose or it no longer holds the BladeFLASK firmly.
Why are there two different colours of the Qlicksmart BladeFLASK?
The Qlicksmart BladeFLASK has been designed to comply with the international standard colours for medical sharps waste. The red unit complies with US, Central America, and South America standards, and the yellow unit complies with most other countries’ standards. Please consult your organization to determine which unit complies with local standard colours for medical sharps waste.
Is the BladeFLASK latex free?
YES. The BladeFLASK body is made of a thermoplastic, acrylonitrile butadiene styrene (ABS) and Acetyl, and the inner mechanism in made from Polyacetal (POM). The Mounting Bracket is made from acrylonitrile butadiene styrene (ABS).
Reviews
Customer Reviews
Products
Other Products

Partner with Qlicksmart & Lead in Safety Innovation
Distribute industry-leading safety solutions and provide professionals with the tools they need to work smarter and safer. Let’s work together to make workplaces safer and more efficient.