Medical Devices ISO 13485:2016
Our Mission to Make Healthcare Safer
Smart Safety Solutions
for Healthcare Professionals
At Qlicksmart, we design simple, effective safety devices to help reduce sharps injuries and improve clinical workflows.
We have over 20 year of R&D experience in the healthcare sector including a worldwide network of distributors.
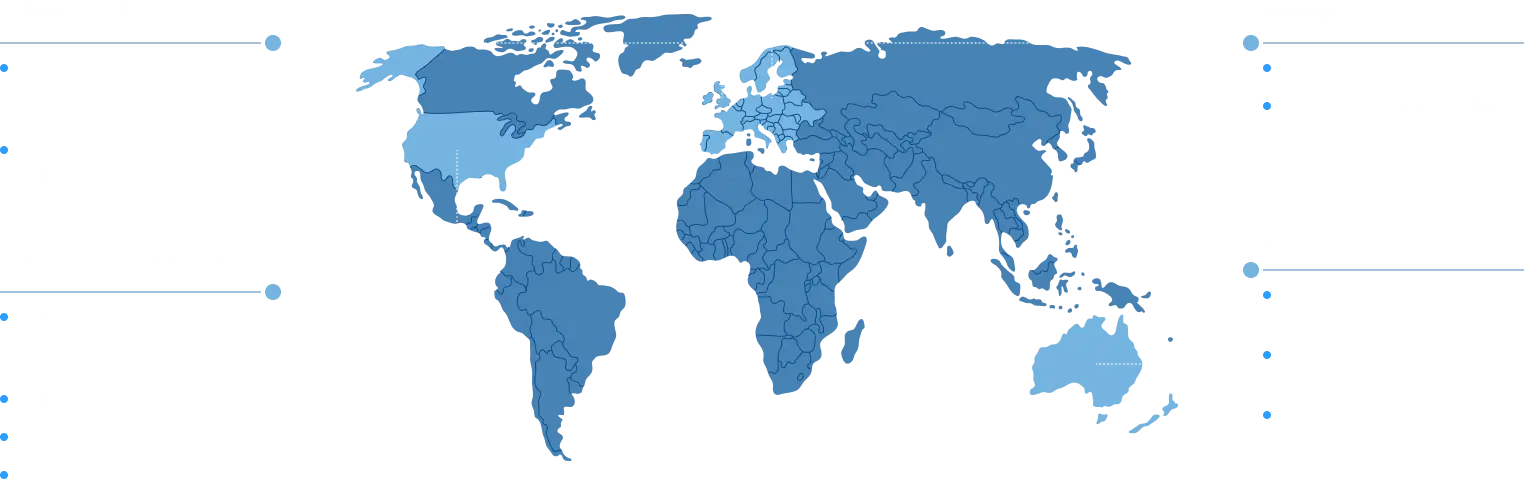
Qlicksmart collaborates with partners in over 50 countries worldwide. We have a wide network of distributors globally.
Recognised with over 20 awards, Qlicksmart’s innovations have been consistently celebrated for advancing safety and excellence in healthcare.
Supporting Qlicksmart’s Global Growth
Smartstream, the Channel Management Company for Qlicksmart products, was established as a response to the growth of Qlicksmart and the recognition of the need for specialist distribution management. This also allowed for Qlicksmart to focus on its continuing R&D, product development and manufacturing supervision.
Qlicksmart was established by two Australian doctors looking for a better way to remove used scalpel blades.


Qlicksmart was founded in 1998 by Australian doctors Neville Henry and Michael Sinnott to address the dangers of scalpel blade injuries. Sparked by a nurse’s frustration, they created the BladeFLASK, the world’s first single-handed blade remover. This success led to the development of sterile solutions like the BladeCASSETTE, BladeNeedleSYSTEM, and BladeSINGLE. Qlicksmart has since expanded into other safety tools like the SnapIT ampoule opener and the CheckCLIP for medication identification, helping protect healthcare workers globally.

Dr. Michael Sinnott is a senior clinician and innovator with over 30 years in emergency medicine. He has trained thousands of doctors, led significant research initiatives, and played a key role in shaping safety standards in Australia and the USA. As a co-founder of Qlicksmart and Staff and Patient Safety, his work continues to improve healthcare safety through research, product innovation, and advocacy.

Qlicksmart today
is a global leader in sharps safety.
Today, we focus on developing smart safety products and maintaining high standards across our processes and suppliers. Our solutions support sharps management and medication labeling in healthcare.
We also promote staff safety through education, advocacy, and collaboration with regulatory bodies to improve hospital safety standards worldwide.
We are committed to fulfilling customer satisfaction and regulatory requirements through quality management systems for continued improvement of our products and services. Qlicksmart is accredited to ISO 13485, the International Standard for Quality Management System for medical device companies. In particular, our quality system complies with:
Awards and Recognitions

Qlicksmart Product Applications Across Industries
Qlicksmart sharp safety solutions are used in over 2,000 healthcare facilities in the USA, nearly 220 hospitals in the UK, and 185 hospitals in Japan, ensuring safer sharps handling across various medical settings.
Qlicksmart sharp safety solutions are used in over 2,000 healthcare facilities in the USA, nearly 220 hospitals in the UK, and 185 hospitals in Japan, ensuring safer sharps handling across various medical settings.
For over 25 years, our efforts in reducing injuries and medication errors have been evidenced in many hospitals, clinics, universities, and schools around the world, including Australasia, Europe, Asia, North and South America. Our aim is not only to create and produce world-class safety devices, but also to inform and educate sharps safety to
all users.
We’ve compiled our learning modules, videos, and tools to help prepare you and your colleagues to a safer working environment. These can be used to enhance learning programs regarding safety concepts, and to provide purchasing departments with background information on regulatory and clinical safety requirements.
Blog
The Latest from Qlicksmart

Risky Business: Operating Theatres

A Smarter, Safer Workplace: How AI is Changing...

The New Standard of Care: Staff Safety

Partner with Qlicksmart & Lead in Safety Innovation
Distribute industry-leading safety solutions and provide professionals with the tools they need to work smarter and safer. Let’s work together to make workplaces safer and more efficient.